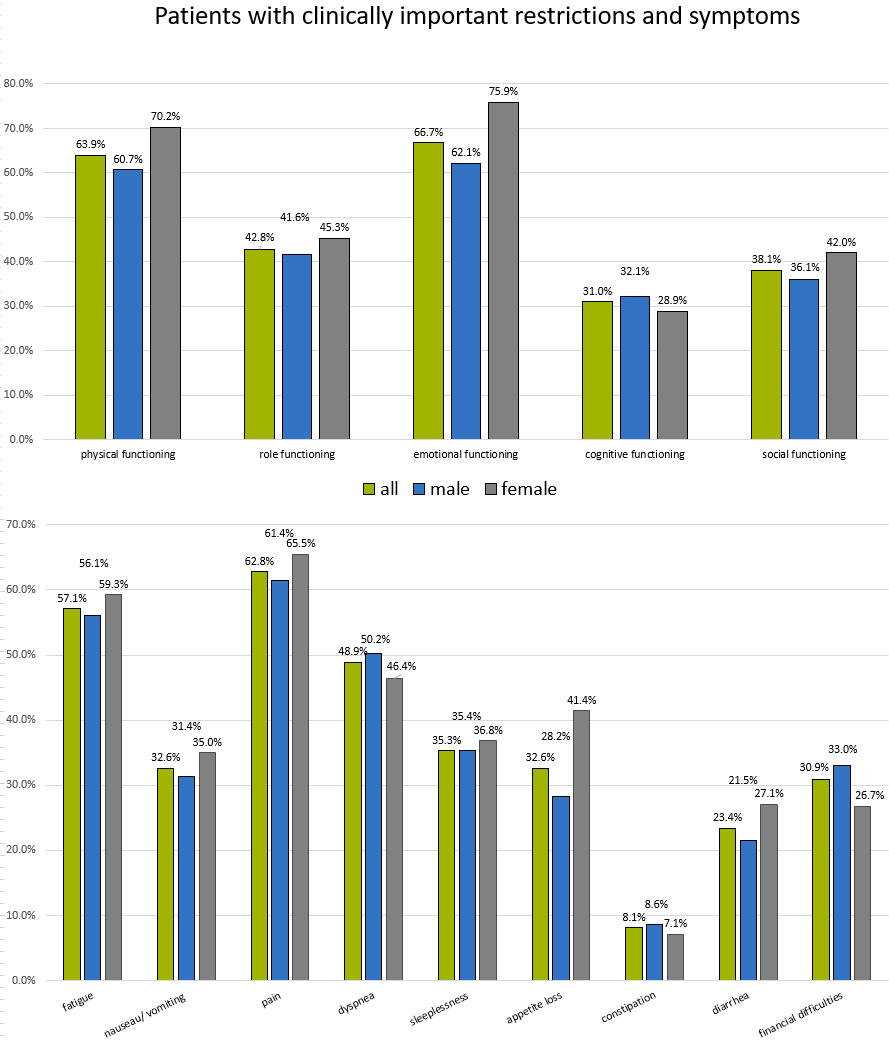
Patient-reported outcomes (PROs) play an important role in determining the health status of tumor patients. They are used to measure various aspects of a patient’s health (physical, mental, social) and are utilized for a wide range of purposes – from clinical trials to everyday care and health services research.
Our core unit is involved in the electronic collection of patient-reported outcomes. The idea is that PROs should be immediately utilizable by healthcare professionals and directly integrated into medical practice. In close cooperation with other departments and institutions we conduct studies that evaluate, develop and use patient-reported outcomes and promote their implementation in healthcare practice.
The Core Unit is an integrated part of the research structure "Oncological Outcome and health-services research".